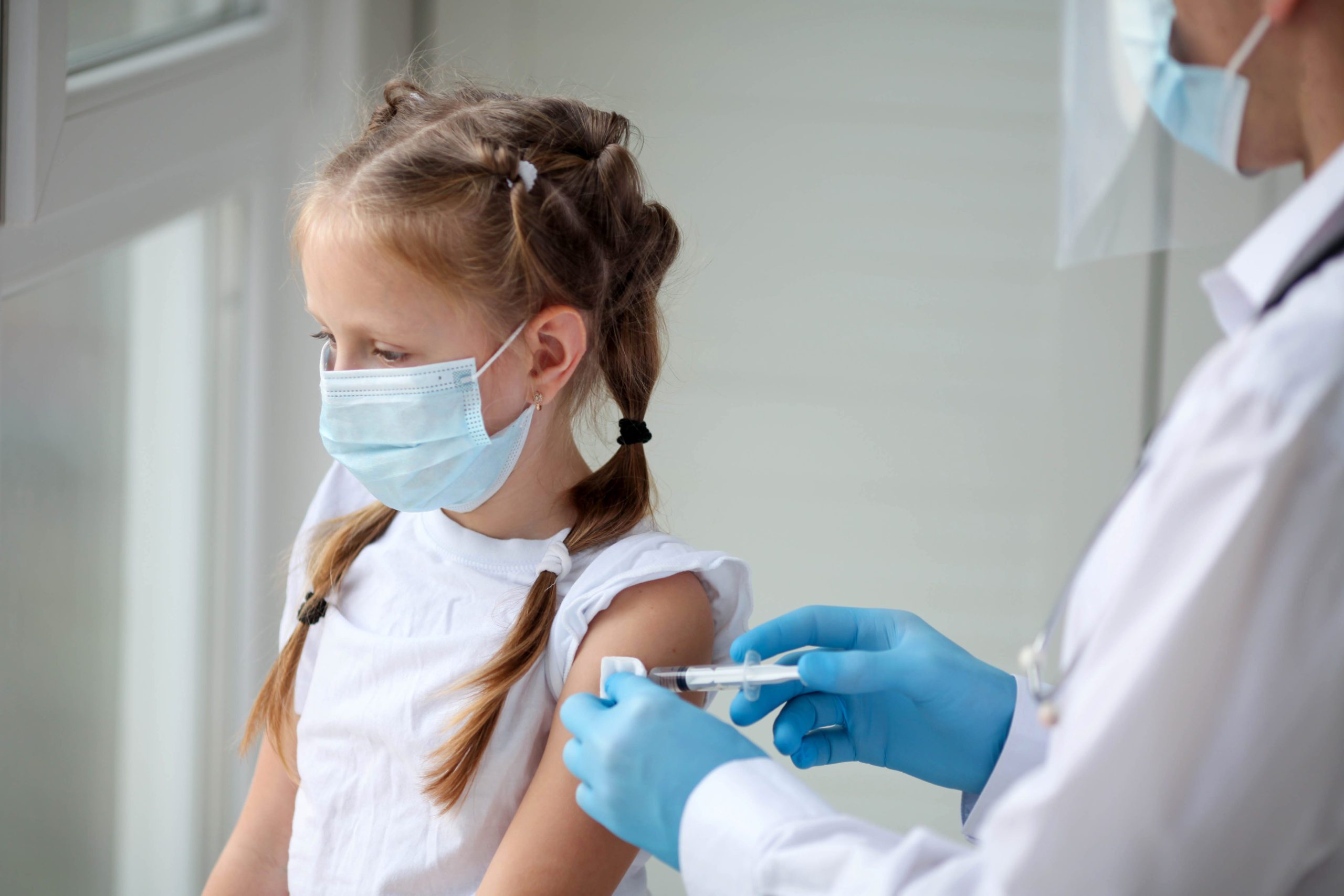
The U.S. Food and Drug Administration (FDA) approved the use of Pfizer-BioNTech and Moderna COVID vaccines for use in children under 5 on Friday.
The move comes after an expert panel brought together by the FDA voted unanimously on Wednesday to recommend the vaccines for that age group, Time reported.
The panel voted 21-0 that the positive effects of the Moderna & Pfizer BioNTech vaccines outweigh the potential side effects for children 6 months to 4 years. This does not automatically mean the vaccines will be made available for public use, but the FDA usually follows the recommendations of its advisory panels.
There is another step in the approval process. Next, the Centers for Disease Control and Prevention (CDC) will meet Saturday to discuss guidelines for pharmacies and doctors’ offices, CNBC reported.
CDC Director Dr. Rochelle Walensky will then need to sign off on that guidance before kids can begin receiving shots.
The White House announced it believes shots could be administered as early as next week.
There are those that disapprove of the measure, though.
On Thursday, Florida Governor Ron DeSantis said he is opposed to administering the vaccines to young kids, Market Watch reported.
“We are affirmatively recommending against doing COVID vax for young kids, particularly the 6-month-old, the 2-year-old little kids,” he said at a press conference.
DeSantis argued that young children are not as susceptible to the virus and the benefits do not outweigh the risks.
“Our department of health has been very clear,” DeSantis said. “These risks outweigh the benefits, and we are recommending against. That isn’t the same as banning it. People can access it if they want to.”
Infection rates in this age group are still relatively low. The latest CDC data shows a 3.3% positive-case rate (about 2.5 million confirmed cases) among children zero to 4 years old as of Thursday morning. Approximately 480 COVID-related deaths have been recorded in this age group since the start of the pandemic, accounting for .01% of all such deaths in the U.S.
The two vaccines give parents a choice. The Moderna vaccine will offer a two-shot regimen, and the Pfizer-BioNTech option will be a three-shot series.
Pfizer-BioNTech changed to the three-shot dose after trials showed a two-shot regimen of its vaccine did not create sufficient immune response.
The FDA released a brief before its Wednesday meeting outlining the results of the Pfizer-BioNTech trials. Preliminary data in the report concluded the vaccine was 80.4% effective in preventing symptomatic COVID-19 in children between 6 months and 5 years old.
Side effects, according to the FDA report, included irritability in children 6 to 23 months old and injection site pain and fatigue for children from 2 to 4.
Last week, the FDA provided details on the Moderna two-shot regimen as well, Time reported. Data indicated that the vaccine was 51% effective in children 6 to 23 months old and 37% effective in children 2 to 5.






It’s been proven that C-19 jabs are dangerous for young and adolescent children. Parents who subject their children to experimental drugs should be arrested for child abuse.
Agreed 100%!
Before you put that mRNA concoction in your children, PLEASE read up on the deaths and injury caused our youth by the jab. Consider this: 1. It is NOT a vaccine. I does NOT prevent the disease. 2. It wll require boosters in 6-9 months. 3. Multiple deaths and other illnesses result from the jab. 4. There are drugs that end the symptoms rapidly – mono clonal antibodies, Ivermectin, etc. 5. If we have anti-viral drugs to limit the disease – WHY is it necessary to put other drugs that rapidly reach the blood brain barrier and no one knows how long it will stay in the brain. 6. Do a little research and discover who is making big bucks over this MAN MADE DISEASE.
This could be a death sentence for your child!!!